Fact or Fiction: PFS to OS is different in immuno-oncology
Whilst there are good reasons to believe this might be true, like all good statisticians I’m sceptical until I see some data. If the relationship indeed were different, there would be important implications for trial design and development strategy– I return to this later.
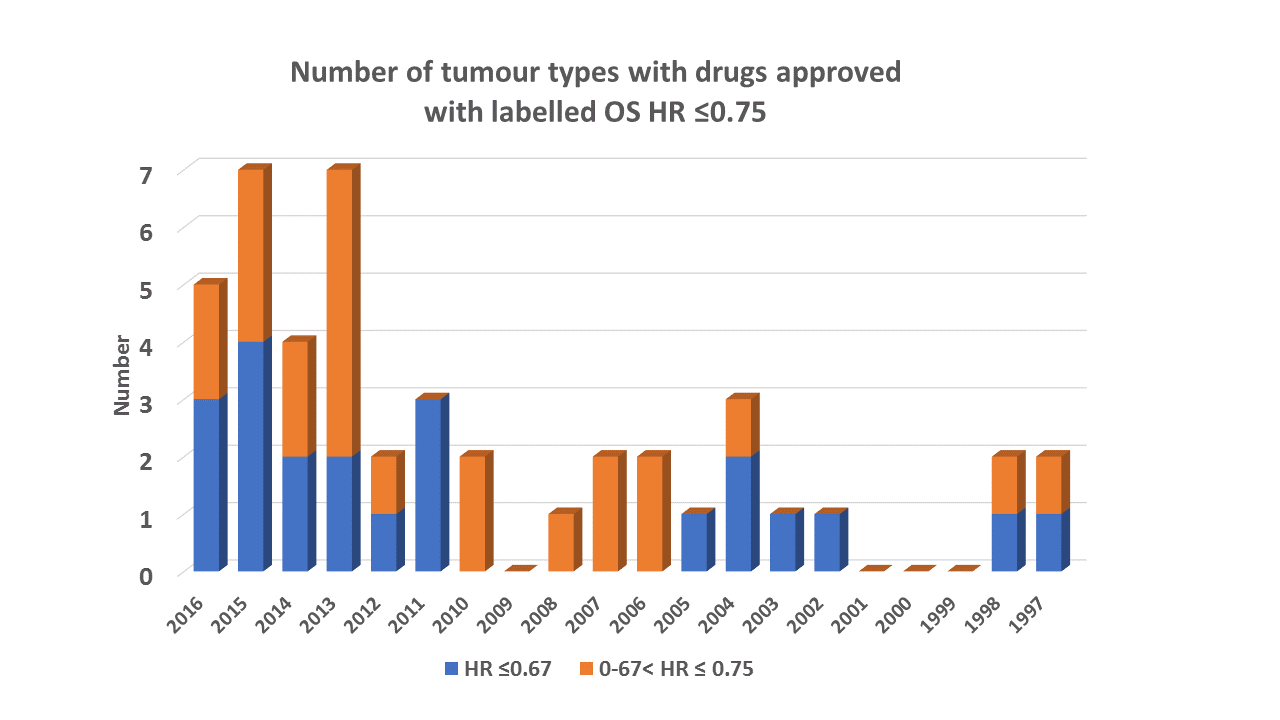
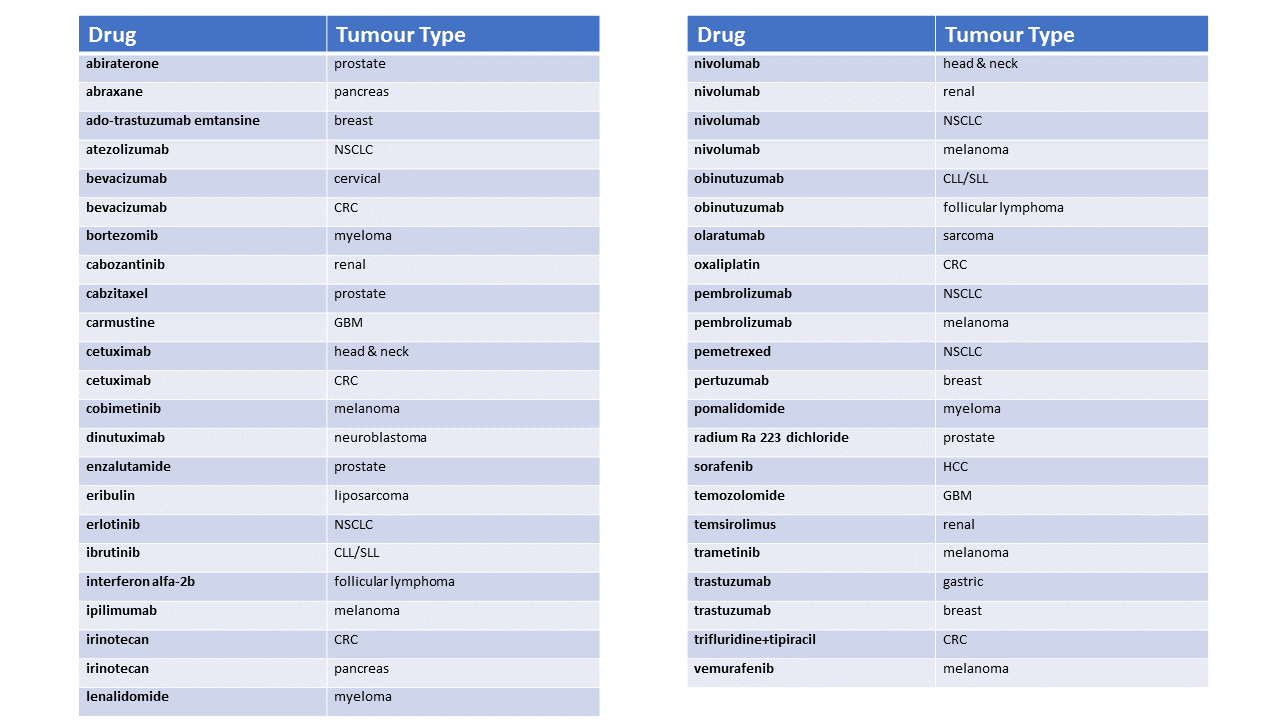
Following the recent ESMO conference there are now plenty of published data. Plotted here, and identified by tumour type, are the published hazard ratios (HRs) for randomised trials with nivolumab, pembrolizumab, atezolizumab and ipilimumab. The size of the bubble is proportional to the number of deaths.

There does indeed seem to be a different relationship: The dashed line shows the line of unity (OS HR = PFS HR), and with previous classes of therapy the points typically would lie above the line, indicating the OS HR was closer to 1. Furthermore, the relationship appears to be different between melanoma and NSCLC why might that be?? And not surprisingly, given these agents have shown OS benefits in later lines of therapy, those trials that experienced crossover from control to immunotherapy (bubbles greyed out, >40% crossover) the OS effect is diluted.
That’s all very interesting but so what??
Well, this is great news if you’re first, or very early, in a tumour type and can perform your trials without crossover, and you hedge your bets between PFS and OS by splitting alpha. However, if you’re later the alpha OS back-stop may well fail due to crossover to agents in the same class.
In terms of trial design you’ll need to consider the following
Do I hedge and keep some alpha for both OS and PFS, assuming PFS is a viable registration endpoint in that setting
What’s the best split of alpha between PFS and OS?
You’ll probably want to plan to analyse OS at the same time as PFS and later, how do I control my alpha spend?
Furthermore, what if you have multiple arms and sub-populations, how do I control my alpha then and still preserve power?
When do I analyse these endpoints given there is often a delayed effect, to avoid the HR being diluted?