Golden age in oncology - methodology
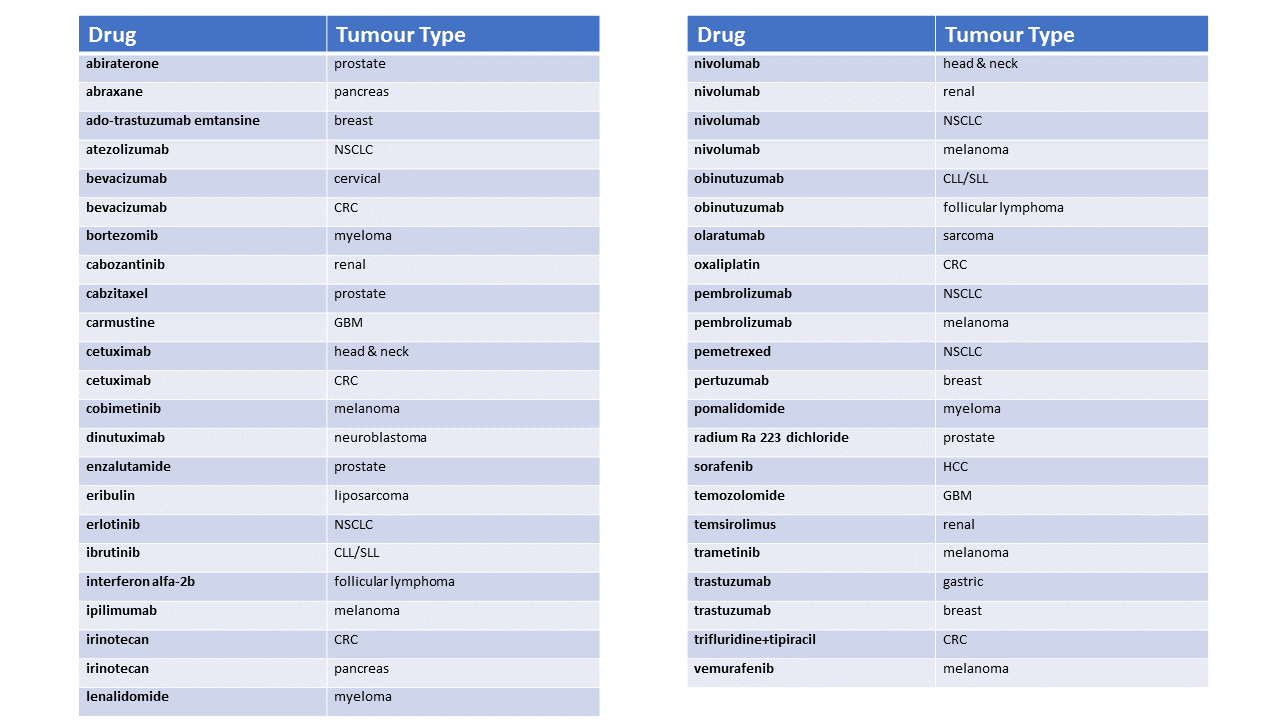
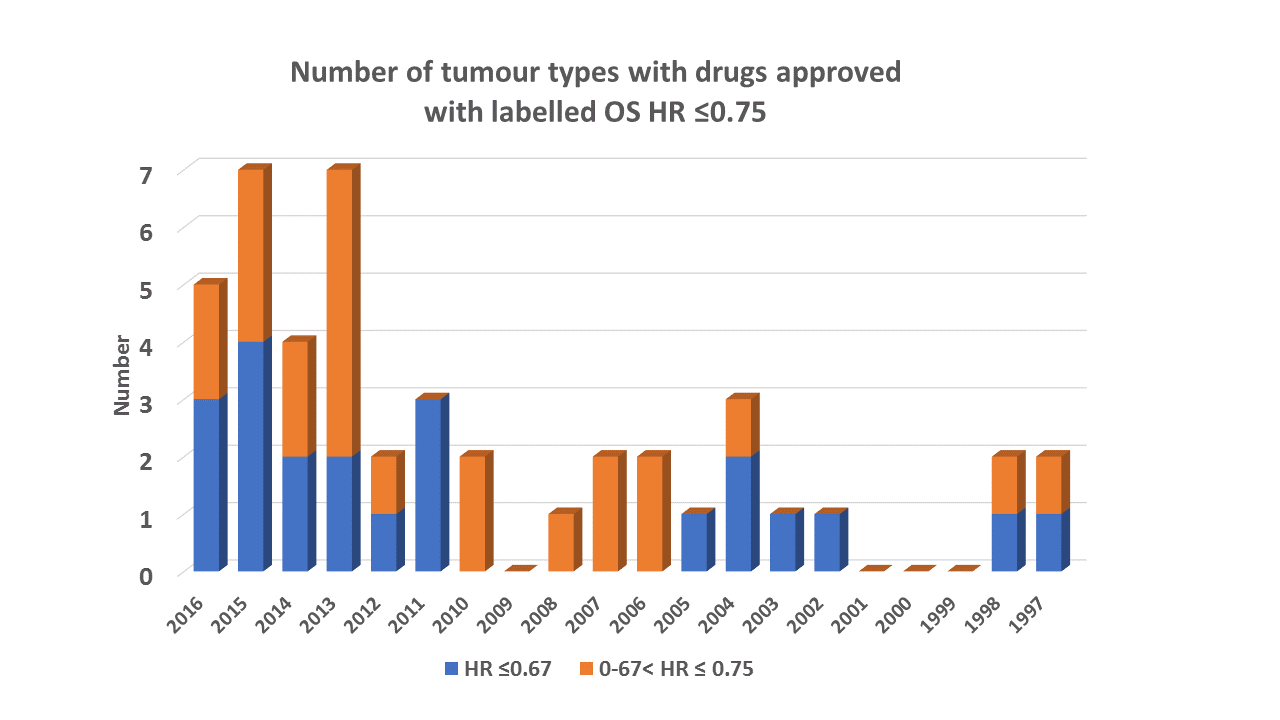
All approved oncology drugs here in advanced cancer indications were identified from here with labels accessed in December 2016.
A drug was counted a maximum of once per tumour type from US labels and only if it was both less than the relevant HR threshold and statistically significant. In some cases the upper confidence limit for the HR was < 1 but the label indicated that the effect was not statistically significant. This occurred in cases where interim analyses were presented but had not crossed the pre-defined stopping boundary. If more than one analysis of OS appears in the label, only the analysis corresponding to the first time the interim stopping boundary was assessed.
If the indication corresponded to a subgroup of the trial population, the OS result in the corresponding subgroup analysis was compared to the thresholds. If both PII and PIII data were presented, only PIII data were considered. If more than one experimental arm was presented, only OS data from the approved dose/regimen was evaluated.
Drugs are assigned to the year in which they were first approved for that tumour type. For example, if a drug was approved on a single arm trial but later showed an OS advantage in the same tumour type, the year assigned was the one when the drug was initially made available in that tumour type. In some older labels only the p-value was available, the FDA statistical review was used to derive the HR.
The full list of drugs with an OS HR ≤ 0.75 from 1997-2016